Background
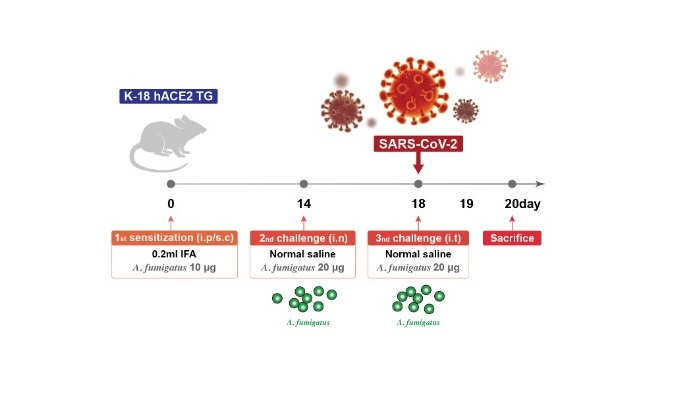
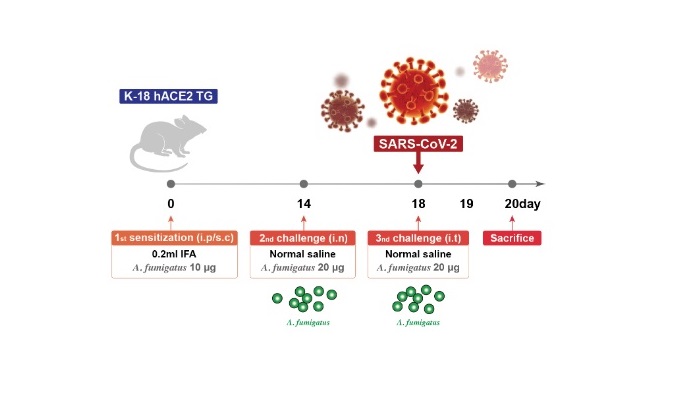
To the Editor,Recent clinical data suggest that the outcome of coronavirus disease 2019 (COVID-19) in asthmatic patients depends on the subtype and severity of asthma1,2; however, there is limited information on the pathobiological outcomes of superimposed SARS-CoV-2 infection in asthma, especially in severe disease.To investigate the effects of SARS-CoV-2 infection on severe asthma, we developed a novel mouse model using SARS-CoV-2- susceptible hACE2 transgenic mice, K18-hACE2.3 Specifically, we first established Aspergillus fumigatus (Af)- induced severe eosin-ophilic allergic lung inflammation using fungal extracts4 and chal-lenged the mice intranasally with SARS-CoV-2 on the same day as the last Af challenge (Figure1A).Forty-eight hours after the last challenge with Af, when fungus-induced eosinophilic airway inflammation predominated, greater in-filtration of inflammatory cells was noted in the peribronchiolar and perivascular areas in the Af- challenged mice after SARS-CoV-2 in-fection. This was supported by the increased number of eosinophils in the bronchoalveolar lavage fluid (BALF) (Figure1B,C). Moreover, in parallel with the increase in neutrophils in BALF, SARS-CoV-2 in-fection led to pronounced infiltration of alveolar inflammatory cells in Af- challenged mice compared with those sensitized only to Af and infected with the virus.Cytokines implicated in severe asthma after SARS-CoV-2 infec-tion were significantly increased in the lung tissues of Af- challenged mice (Figure 1D). In mice sensitized only to Af, S A R S – C o V – 2 i n f e c-tion did not induce hyperinflammatory changes in the lungs at 48 h, when they were in the early stages of infection.3 However, the pulmonary levels of the hyperinflammation markers of COVID-19 were dramatically increased in Af- challenged mice 48 h after viral infection (Figure1E). Experiments using the delta variant B.1.617.2 [NCCP43390] showed similar findings (Figure1F,G).The NLRP3 inflammasome was recently reported to play a cru-cial role in the pathobiology of COVID-19.5 We analyzed six public datasets of gene expression profiles of airway and blood specimens from COVID-19 patients and found that the expression of NLRP3 inflammasome-related genes was significantly higher in airway specimens than in blood samples (Figure S1A,B). Furthermore, by analyzing a single-cell RNA sequencing dataset,6 we characterized several clusters of macrophages and dendritic cells harboring acti-vated NLRP3 inflammasome in nasal swab samples from patients with early COVID-19 (Figure S1C,D).Given the crucial role of the NLRP3 inflammasome in the patho-genesis of current Af- induced severe asthma,4 we investigated whether pre-existing asthmatic inflammation influenced SARS-CoV-2- induced NLRP3 inflammasome activation in the lungs of mice. Notably, significant increases in NLRP3 inflammasome components (NLRP3, ASC, cleaved caspase-1) and mature IL-1β were observed after SARS-CoV-2 infection in Af- challenged mice compared with Af- challenged mice that had not been infected with SARS-CoV-2 or SARS-CoV-2- infected mice that had not been challenged with Af (Figure2A,B). As expected, blockade of IL-1β remarkably re-duced SARS-CoV-2- induced pulmonary inflammation in these mice (Figure 2C), implying that SARS-CoV-2 infection in severe asthma is associated with worsening of COVID-19 and asthmatic inflammation through NLRP3 inflammasome activation. Transcriptome analyses of the lung tissue from mice and a transcriptome-based predicted gene interaction network analysis further validated these findings (Figure2D,E). In a population-based large nationwide cohort, we also observed a higher severity of COVID-19 in asthma patients re-quiring systemic corticosteroid administration (Figure S2).In conclusion, SARS-CoV-2 infection in severe asthma may be associated with worsening of COVID-19 through respiratory NLRP3 inflammasome activation. Targeting the NLRP3 inflammasome may be a promising approach for early treatment of COVID-19 in a specific molecular phenotype of severe asthma including fungus- induced severe eosinophilic allergic subtype associated with a dys-regulated innate immune response.
Jae Seok Jeong,Jin Young Choi,Jong Seung Kim,Seong Ok Park,Wankyu Kim,Yeo-Gha Yoon,Hae Jin Park,Kyung Hwa Park,Doo Hwan Kim,Jung Mo Kim,Gou Young Koh,Seong Kug Eo,Yong Chul Lee, “SARS-CoV-2 infection in severe asthma is associated with worsening of COVID-19 through respiratory NLRP3 inflammasome activation”, wiley online library. First published: 24 July 2022 PMID: 35871401 PMCID: PMC9349818 DOI: 10.1111/all.15452 (IF 6.655)